On October 19 the Food and Drug Administration (FDA) proposed in a new rule that would pave the way to make available a new category of over-the-counter hearing device, after the 90-day public comment period is complete.
The intention is to change today’s regulatory framework that classifies hearing aids as “medical devices” requiring a prescription. Such regulations add cost and creates barriers for many of the 38 million Americans who suffer some sort of hearing loss, but the new rule would change all of that.
When you can’t hear, you are distanced from connecting with those around you. This distancing, particularly in older adults, can have effects not only of isolation, but also of cognitive decline, according to Dr. Frank Lin of Johns Hopkins University School of Medicine: “The greater the hearing loss, the greater the risk of loss of thinking and memory abilities over time.”
Hearing loss is complex, and there is an important role for hearing tests and a hearing aid prescribed by an audiologist. But Medicare and private insurance typically do not cover the cost of a hearing aid which costs, on average, around $5,000. And that’s a significant barrier, particularly for lower income people and those living in rural areas where access to audiologists may be limited.
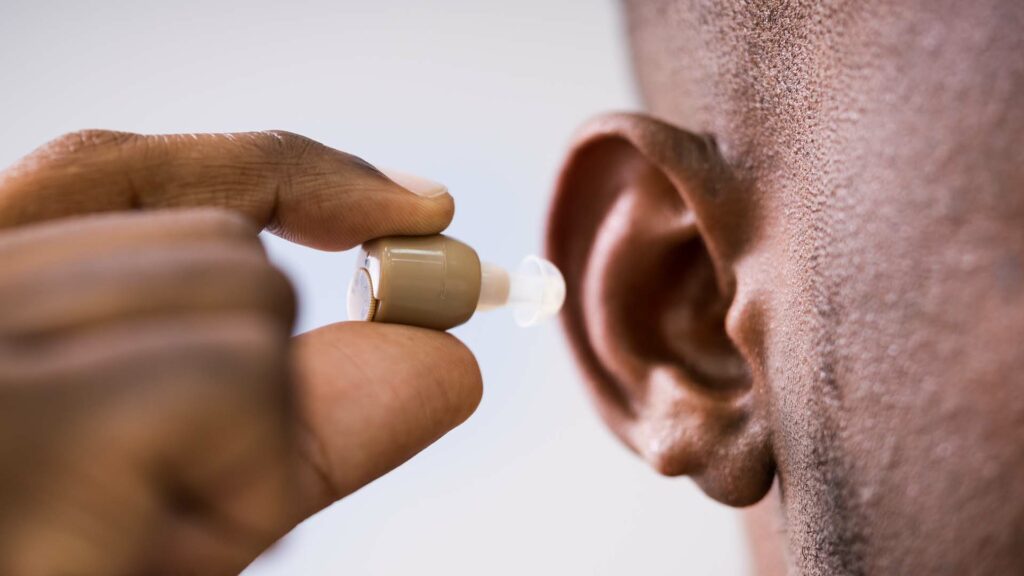
Today there are devices sold over-the-counter called “personal sound amplification products” or PSAPs. These cost around $300; they can only be marketed as a tool to amplify sound—to make things like music louder—for people with normal hearing. But there is no way for consumers to know if they are safe or effective.
This new FDA rule would bring quality control to a new hearing-aid category for those with mild hearing loss. You’ll likely have to wait until next summer to purchase a hearing aid over the counter, but this makes it a promising reality.